Year :
1983
Title :
Chemistry
Exam :
JAMB Exam
Paper 1 | Objectives
41 - 50 of 50 Questions
| # | Question | Ans |
|---|---|---|
| 41. |
A jet plane carrying 3,000 kg of ethane burns off all the gas forming water and carbondioxide. if all the carbondioxide is expelled and all the water formed is condensed and kept on board the plane, then the gain in weight is A. 1,800 kg B. 900 kg C. 600 kg D. 2, 400 kg E. 1, 200 kg |
D |
| 42. |
Liquid X, reacts with sodium trioxocarbonate (IV) (Na2CO3) to give a gas which turns calcium chloride solution milky X is A. Na2SO4(aq) B. Kl (aq) C. an alkali D. an acid E. a hydrocarbon |
D |
| 43. |
Which of the following statements is FALSE? A. Copper (ll) ion can be reduced to copper (l) ion by hydrochloric acid and zinc B. Sodium metal dissolves in water giving oxygen C. Nitrogen is insoluble in water D. Carbondioxide is soluble in water E. lead has a higher atomic weight than copper |
B |
| 44. |
When sodium dioxonitrate (lll) (NaNO2) dissolves in water, ∆H is positive. The process of dissolution is A. exothermic B. endothermic C. isothermal D. isomeric E. hygroscopic |
B |
| 45. |
The equilibrium reaction between copper (l) chloride and chlorine at 25oC and 1 atmosphere is represented by the equation: A. more CuCl is formed at 40oC B. more CuCl2 is formed at 10oC C. Less CuCl2 is formed at 10oC D. there is no change in the amount of CuCl2 formed at 40oC and10oC E. more CuCl is consumed at 40oC |
B |
| 46. |
Zn + 2HCl → ZnCl2 + H2 A. the zinc is in the powered form B. a greater volume of the acid is used C. a smaller volume of the acid is used D. the reaction vessel is immersed in an ice-bath E. the zinc is in the form of pellets |
A |
| 47. |
Zn + H2 SO4 → ZnSO4 + H2 A. 1.35g B. 1.00g C. 0.70g D. 0.65g E. 0.00g |
A |
| 48. |
30 cm3 of 0.1 M Al (NO3)3 SOLUTION IS RECTED WITH 100cm3 of 0.15M of NaOH solution. Which reactant is in excess, and by now much ? A. NaOH solution by 70 cm3 B. NaOH solution, by 60 cm3 C. NaOH solution by 40 cm3 D. Al(NO3)3 solution by 20 cm3 E. Al(NO3)3 solution by 10 cm3 |
C |
| 49. |
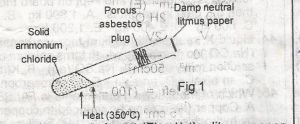 In the above experiment, the litmus paper will initially A. be bleached B. turn green C. turn red D. turn blue E. turn black |
D |
| 50. |
 In the experiment above, a current was passed for 10 minutes and 0.63g of copper was found to be deposited on the cathode of CuSO4 cells. The weight of silver deposited on the cathode og AgNO3 cell during the same period would be? [Cu = 63, Ag = 108] A. 0.54g B. 1.08g C. 1.62g D. 2.16g E. 3.24g |
B |
| 41. |
A jet plane carrying 3,000 kg of ethane burns off all the gas forming water and carbondioxide. if all the carbondioxide is expelled and all the water formed is condensed and kept on board the plane, then the gain in weight is A. 1,800 kg B. 900 kg C. 600 kg D. 2, 400 kg E. 1, 200 kg |
D |
| 42. |
Liquid X, reacts with sodium trioxocarbonate (IV) (Na2CO3) to give a gas which turns calcium chloride solution milky X is A. Na2SO4(aq) B. Kl (aq) C. an alkali D. an acid E. a hydrocarbon |
D |
| 43. |
Which of the following statements is FALSE? A. Copper (ll) ion can be reduced to copper (l) ion by hydrochloric acid and zinc B. Sodium metal dissolves in water giving oxygen C. Nitrogen is insoluble in water D. Carbondioxide is soluble in water E. lead has a higher atomic weight than copper |
B |
| 44. |
When sodium dioxonitrate (lll) (NaNO2) dissolves in water, ∆H is positive. The process of dissolution is A. exothermic B. endothermic C. isothermal D. isomeric E. hygroscopic |
B |
| 45. |
The equilibrium reaction between copper (l) chloride and chlorine at 25oC and 1 atmosphere is represented by the equation: A. more CuCl is formed at 40oC B. more CuCl2 is formed at 10oC C. Less CuCl2 is formed at 10oC D. there is no change in the amount of CuCl2 formed at 40oC and10oC E. more CuCl is consumed at 40oC |
B |
| 46. |
Zn + 2HCl → ZnCl2 + H2 A. the zinc is in the powered form B. a greater volume of the acid is used C. a smaller volume of the acid is used D. the reaction vessel is immersed in an ice-bath E. the zinc is in the form of pellets |
A |
| 47. |
Zn + H2 SO4 → ZnSO4 + H2 A. 1.35g B. 1.00g C. 0.70g D. 0.65g E. 0.00g |
A |
| 48. |
30 cm3 of 0.1 M Al (NO3)3 SOLUTION IS RECTED WITH 100cm3 of 0.15M of NaOH solution. Which reactant is in excess, and by now much ? A. NaOH solution by 70 cm3 B. NaOH solution, by 60 cm3 C. NaOH solution by 40 cm3 D. Al(NO3)3 solution by 20 cm3 E. Al(NO3)3 solution by 10 cm3 |
C |
| 49. |
 In the above experiment, the litmus paper will initially A. be bleached B. turn green C. turn red D. turn blue E. turn black |
D |
| 50. |
 In the experiment above, a current was passed for 10 minutes and 0.63g of copper was found to be deposited on the cathode of CuSO4 cells. The weight of silver deposited on the cathode og AgNO3 cell during the same period would be? [Cu = 63, Ag = 108] A. 0.54g B. 1.08g C. 1.62g D. 2.16g E. 3.24g |
B |